Michael Allen and Gumpon Prateepchaikul
Department of Mechanical Engineering, Faculty of Engineering, Prince of Songkla University, Hadyai, Thailand
Download the "Macromodel" MS Excel spreadsheet (for Windows) -- 320kb
D. Darnoko and M. Cheryan: Kinetics of Palm Oil Transesterification in a Batch Reactor (72kb Acrobat pdf)
H. Noureddini and D. Zhu: Kinetics of Transesterification of Soybean Oil (540kb Acrobat pdf)
Abstract
Introduction
Reversible reactions
Methodology
Results
Discussion
Conclusions
References
Figures
Abstract
The design of a reactor for the production of palm-oil methyl ester required the modelling of the complex series of reversible reactions which are known to take place. Rate equations were derived which could be solved by a number of mathematical techniques: In this case, a finite-difference technique was used based on simple spreadsheet functions. The resulting model was compared with results published in the literature as well as with results obtained in the Department of Mechanical Engineering of the Prince of Songkla University. Applying this model made possible several recommendations to enhance palm-oil methyl ester production in batch reactors of the form currently operated by Energy Group of the Prince of Songkla University.
Introduction
Palm oil consists of the glycerides of long-chain fatty acids. The carbon chain in these fatty acid residues are comprised of an even number of carbon atoms (typically from 12 atoms (lauric acid) to 20 atoms (arachidic acid) in length) with roughly half of the oil molecules showing some unsaturated carbon-to-carbon bonds. Typically nine major fatty acid residues are found in the glycerides which make up palm oil (see Table 1). Because glycerol is a trihydric alcohol, up to three of these fatty acid residues can be attached to the glycerol structure. But there is no reason to suppose that the three attached to any one molecule are identical.
There are therefore many different molecules available for the reaction with methanol in the transesterification reaction which produces the palm-oil methyl esters (POME). And it can be anticipated that due to variable saturation, variable chain length and steric hindrance within the glyceride molecule that each reacts at a slightly different rate. In applying methods more usually applied to the design of chemical reactors to synthesise pure chemicals from similarly pure chemicals, it is immediately apparent that there is a lack of certainty as to which reaction is proceeding and what precisely is controlling it. This has not, however, discouraged the production of palm-oil methyl ester: This is because palm-oil methyl ester is one of the most successful sustainable and bio-degradable fuel oils ever synthesised from a natural product.
(adapted from Darnoko & Cheryan [1])
Palm-oil methyl ester is one of a series of "biodiesels" formed from natural oils and tested in vehicles in Europe, the United States and, more recently, in Thailand. It is reported to produce no sulphur dioxide, less carcinogenic fumes and less black smoke. The lack of sulphur in the fuel means that it can meet all international standards for fuels suitable for use with catalytic afterburners. It has already been extensively used to dilute conventional petro-diesel and been shown to be biodegradable. Indeed, it has been used to assist in cleaning up "petro-oil" spills. Environmentalists also like it because the carbon dioxide it produces on burning will be absorbed naturally and recycled without an overall net increase in the atmospheric CO2 inventory. In addition, the exhaust fumes from an engine running on palm-oil methyl esters have a smell comparable with pleasant cooking smells. Several countries (France and Germany in particular) have encouraged the widespread use of "biodiesel" blends with conventional "petrodiesel". Indeed it is doubtful that most countries will be able to grow sufficient oil-producing plants to meet the expected demand. In this respect, Thailand is extremely fortunate in that it has a well-established palm-oil industry. It also happens that the oil-palm produces more than twice as much oil per hectare as any other oil-producing crop.
To meet the expected domestic and international demand will require efficient and clean chemical reactors to process the palm-oil into the methyl ester.
Reversible reactions
The complicated chemistry of the reaction is due to the various groups attached to the glycerol molecule. We can use R1, R2, R3 etc to denote that these groups can all have different molecular lengths. Thus a triglyceride can be shown to react with methanol to produce diglycerides, the diglycerides react with another methanol molecule to produce monoglycerides and then these react with a third methanol molecule to produce glycerol. (See Figure 1.)
At each of these three stages, a molecule of fatty acid methyl ester is produced so that, overall, three molecules of methanol are needed to produce three molecules of ester and one molecule of glycerol (see Figure 2).
Each of these reactions is reversible with a different rate constant (kn) denoting that the forward and reverse reactions take place at different rates.
The reactions are all known to be second order (or pseudo-second order) so that we can express the rate of disappearance/appearance of the various components in a series of differential equations (see Figure 3).
Methodology
Several techniques are available for the solution of these differential equations including the use of proprietary software and simulation packages. If a value of ![]() can be chosen that is suitably small, a spreadsheet can be used to predict the molar concentration of each of the major components in terms of their separate concentrations a time interval
can be chosen that is suitably small, a spreadsheet can be used to predict the molar concentration of each of the major components in terms of their separate concentrations a time interval ![]() before. Thus we set the initial concentrations at time 0 and estimate the concentration one second later using the equations presented in Figure 3 as if
before. Thus we set the initial concentrations at time 0 and estimate the concentration one second later using the equations presented in Figure 3 as if ![]() were equivalent to
were equivalent to ![]() .
.
In this present work on the transesterification of palm-oil, an interval of ![]() second was found to give reasonable results.
second was found to give reasonable results.
Although Microsoft Excel was used as the spreadsheet, the technique is valid for any comparable spreadsheet program. No attempt was made in this present study to "fit" experimental curves to the model so that values of k1, k2 etc. could be estimated. Instead, published values of the various rate constants were used. Thus Noureddini and Zhu [2] have published values of k for the transesterification of soybean oil and Darnoko and Cheryan [1] give a more restricted set for the transesterification of palm-oil. Both these authors [1,2] present activation energies so that values of the rate constant can be estimated at different temperatures using the Arrhenius equation k = Ae-E/RT. In addition, Noureddini has determined the effect of varying agitation on both the rate constants k and the energy of activation E.
These k and E values for 50 deg C are presented in Table 2.
It should be remembered that the values of k provided by Darnoko & Cheryan [1] are for a different reaction than the values presented by Noureddini & Zhu [2]. For this reason, exact comparisons are not possible.
Using these values of the activation energy, it was possible to predict the effect of temperature on the various chemical reactions taking place. Various program items were written in VisualBasic to facilitate this within the Excel spreadsheet.
Results
Figure 4 shows the predicted composition during the transesterification of soybean oil at 50 deg C using the k values provided by Noureddini [1]. Figure 5 shows the comparable values at 65 deg C: Both of these results are for a molar ratio of 6:1 for methanol:oil. Figure 6 shows the effect of increasing this molar ratio of methanol:oil to 9:1. Figure 7 and Figure 8 show comparable results using the Darnoko & Cheryan k data derived from their work with palm oil [1]. (The molar values for the mono- & di-glycerides should be read from the secondary y axis on the right of the figure).
Figure 9 shows some preliminary results obtained by the authors.
Discussion
Comparison between Figures 4 and 5 show that the model predicts an increase from 2.48 moles of methyl ester to 2.59 moles by raising the temperature at which the reaction is carried out from 50 deg C to 65 deg C. This represents an increase in percentage conversion from 82.7 to 86.4. There is also a corresponding time reduction to achieve equilibrium from 2000 secs to 1200 secs which would greatly affect the design of a reactor to produce methyl ester.
Comparison between Figures 5 and 6 indicates that an increase in the molar proportion of methanol to oil from 6 to 9 could cause an increase in ester yield from 2.59 moles to 2.86 moles at 65 deg C. This corresponds to an increase in percentage conversion from 86.4 to 95.5. There is a corresponding drop in the time required to achieve equilibrium from 1200 secs to 600 secs which would, in effect, lead to the doubling in the capacity of an existing reactor.
Darnoko & Cheryan [1], provide data for only the forward reactions (k1, k2,and k3) and suggest that they are only valid for the first 1200 secs at 50 deg C. They presumed that thereafter the reaction would be pseudo 1st or even zero order. Figures 7 and 8 reflect this assumption in that they indicate that 100% conversion can eventually be achieved. This is unlikely because the chemical reactions reported are essentially identical with those reported by Nouredinni and Zhu [2] even though the mix of glycerides is different in soybean oil. Comparison of Figures 7 and 8 shows that an increased methanol concentration is predicted to reduce the time required to achieve any degree of conversion just as it does in the results presented in Figures 5 and 6. Another difference is that the model using Darnoko & Cheryan's k values predicts a higher peak concentration of the di- and mono-glyceride than is predicted by the comparable model using Nouredinni and Zhu's values.
Figure 9 shows some preliminary experimental results of a series of trials carried out in a five-litre reactor designed to optimise methyl ester yield from palm oil. The results are presented here in the form of a surface graph which clearly reveals the increased ester formation which is obtained from using an increased methanol:oil ratio in the reactant mix: At a molar ratio of 7.2 moles of methanol per mole of oil, 97.21% of the washed product was found to be methyl ester. This exceeds the current EU specification for 96% ester. The remaining 2.8% comprised unreacted tri-, di- and mono-glycerides. (7.2 molar ratio corresponds with 30% of the oil volume being added as methanol.)
The effect of temperature however, was not as predicted by the spreadsheet model: The experimental work actually shows a decreased methyl ester yield at temperatures above 60 deg C. One possible explanation for this is the loss of excess methanol as a liquid in the reacting mass as its boiling point (64.8 deg C at 1 bar) is approached. Even though there is an efficient condenser fitted to the 5 litre reactor which captures and recycles methanol vapour, there could still be a liquid methanol depletion in the reacting liquid mass.
Another alternative is that the "least squares" method of extracting k values from experimental work is in error in the non-linear system of equations presented as Figure 3. Thus it is possible that several alternative sets of k values could "fit" the observed data. This point was made by Nouredinni and Zhu in their paper [2]. Obviously more experimental work must be done before the reason for this apparent anomaly can be found.
Conclusions
Useful finite-difference models can be easily constructed using a spreadsheet. These models can be enhanced by the incorporation of macros and programs using the Visual Basic language.
The effect of varying initial concentrations, continuous removal of products, dynamic separation of glycerol, temperatures etc., can all be conveniently tested using such a model.
The complete validation of such a model requires further experimental work to understand the nature and origin of apparent anomalies.
References
[1] D. Darnoko and M. Cheryan: Kinetics of Palm Oil Transesterification in a Batch Reactor, JAOCS, Vol. 77, No. 12, pp. 1263-1267. (2000) (72kb Acrobat pdf)
[2] H. Noureddini and D. Zhu: Kinetics of Transesterification of Soybean Oil, JAOCS, Vol. 74, No. 11, pp. 1457-1461. (1997) (540kb Acrobat pdf)
Figures
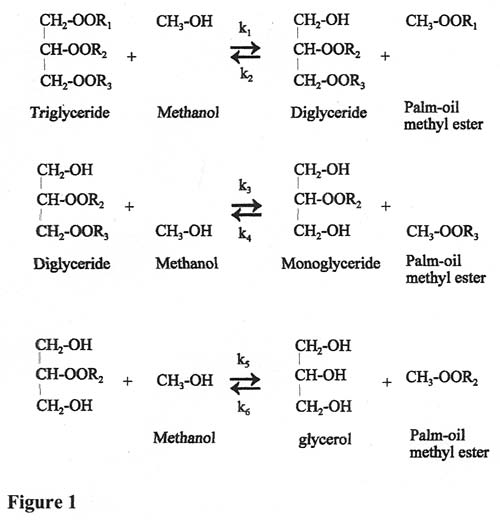
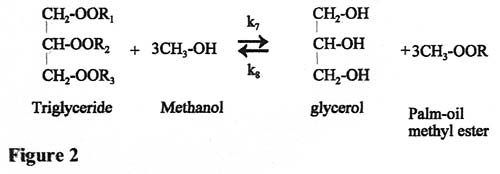

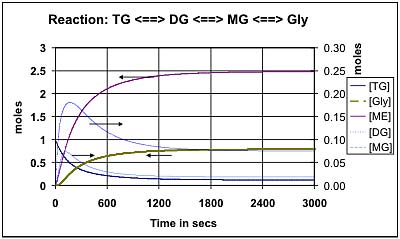
Figure 4 - Transesterification of soybean oil at 50 deg C with a molar ratio of 6:1 for methanol:oil
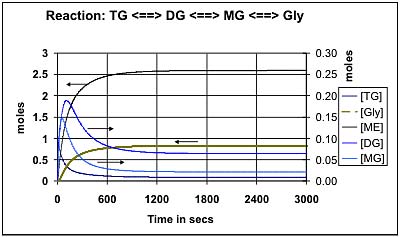
Figure 5 - Transesterification of soybean oil at 65 deg C with a molar ratio of 6:1 for methanol:oil

Figure 6 –Transesterification of soybean oil at 65 deg C with a molar ratio of 9:1 for methanol:oil
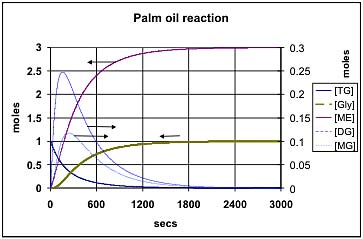
Figure 7 — Transesterification of palm-oil at 65 deg C using a molar ratio of 6:1 for methanol:oil
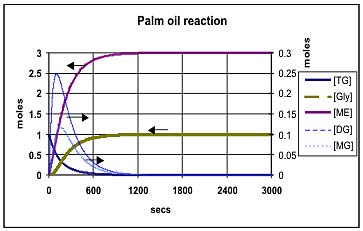
Figure 8 — Transesterification of palm oil at 65 deg C with a molar ratio of 9:1 for methanol:oil
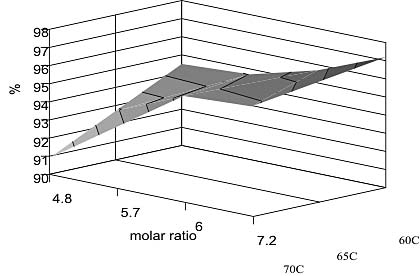
Figure 9 — Percentage conversion of palm oil as a function of temperature and methanol:oil ratio
Back to Biofuels Library
Back to Straight vegetable oil as diesel fuel: References
Biofuels
Biofuels Library
Biofuels supplies and suppliers
Biodiesel
Make your own biodiesel
Mike Pelly's recipe
Two-stage biodiesel process
FOOLPROOF biodiesel process
Biodiesel processors
Biodiesel in Hong Kong
Nitrogen Oxide emissions
Glycerine
Biodiesel resources on the Web
Do diesels have a future?
Vegetable oil yields and characteristics
Washing
Biodiesel and your vehicle
Food or fuel?
Straight vegetable oil as diesel fuel
Ethanol
Ethanol resources on the Web
Is ethanol energy-efficient?
